
Genie 235 BCC Business – Calculadora de 12 dígitos (Dual Power (Solar y batería), Incluye rastrillo vía Pantalla) Plata/Gris : Amazon.es: Oficina y papelería

Genie 235 BCC Business – Calculadora de 12 dígitos (Dual Power (Solar y batería), Incluye rastrillo vía Pantalla) Plata/Gris : Amazon.es: Oficina y papelería

Niobium has a density of 8.57 g/cm3 and crystallizes with the body-centered cubic unit cell. Calculate the radius of a niobium atom - Chemistry Stack Exchange
Calculation of the equation of the state for (top) BCC Fe and (bottom)... | Download Scientific Diagram

Sodium crystallises in b.c.c unit cell. Calculate the approximate number of unit cells in 9.2 g of solium (Atomic mass of Na = 23u).

An element having BCC crystal structure has atomic mass 50 amu and the density is 6.81 g cm ^-3 . Calculate the edge length (in pm) of unit cell.

Calculation of Packing Efficiency in Body-Centered Cubic Structures (BCC) Video | CBSE 12 Solid State - Brilliant Qatar | Brilliant Group of Institutions

Lithium has a BCC structure. Its density is 530 kg m^-3 and its atomic mass is 6.94 g mol^-1 . Calculate the edge length of a unit cell of Lithium metal: ( NA = 6.02 × 10^23 mol^-1 )



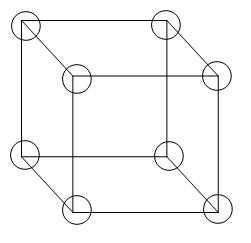

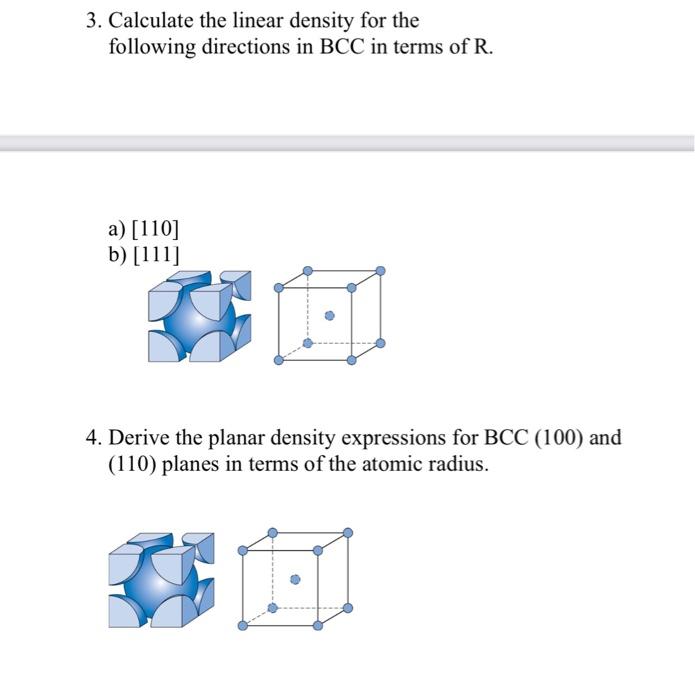

![Solved Calculate the density of the [201] in BCC. (Input | Chegg.com Solved Calculate the density of the [201] in BCC. (Input | Chegg.com](https://media.cheggcdn.com/media/9e3/9e3bf38a-57b2-43e1-b32d-90239acce684/phpaXqS1L)



