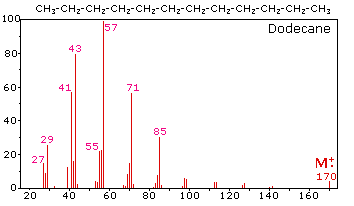
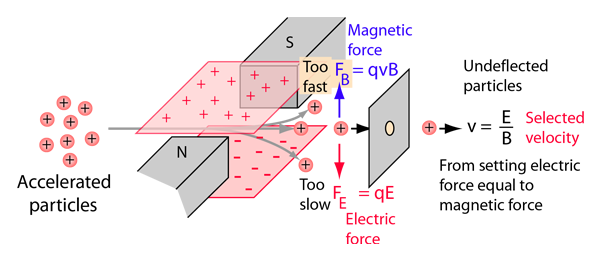
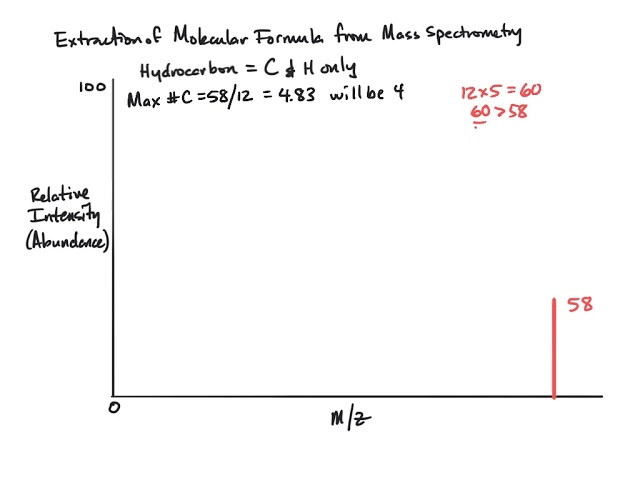
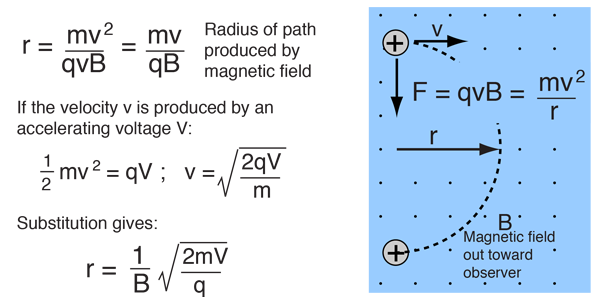
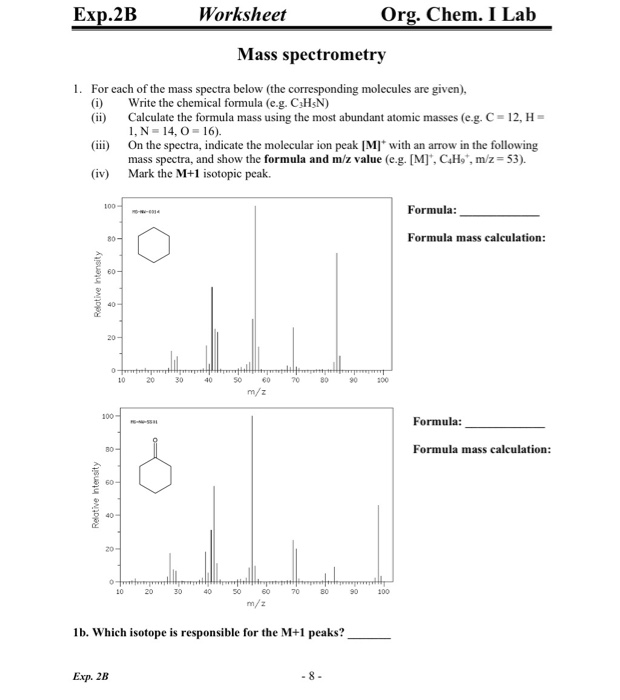
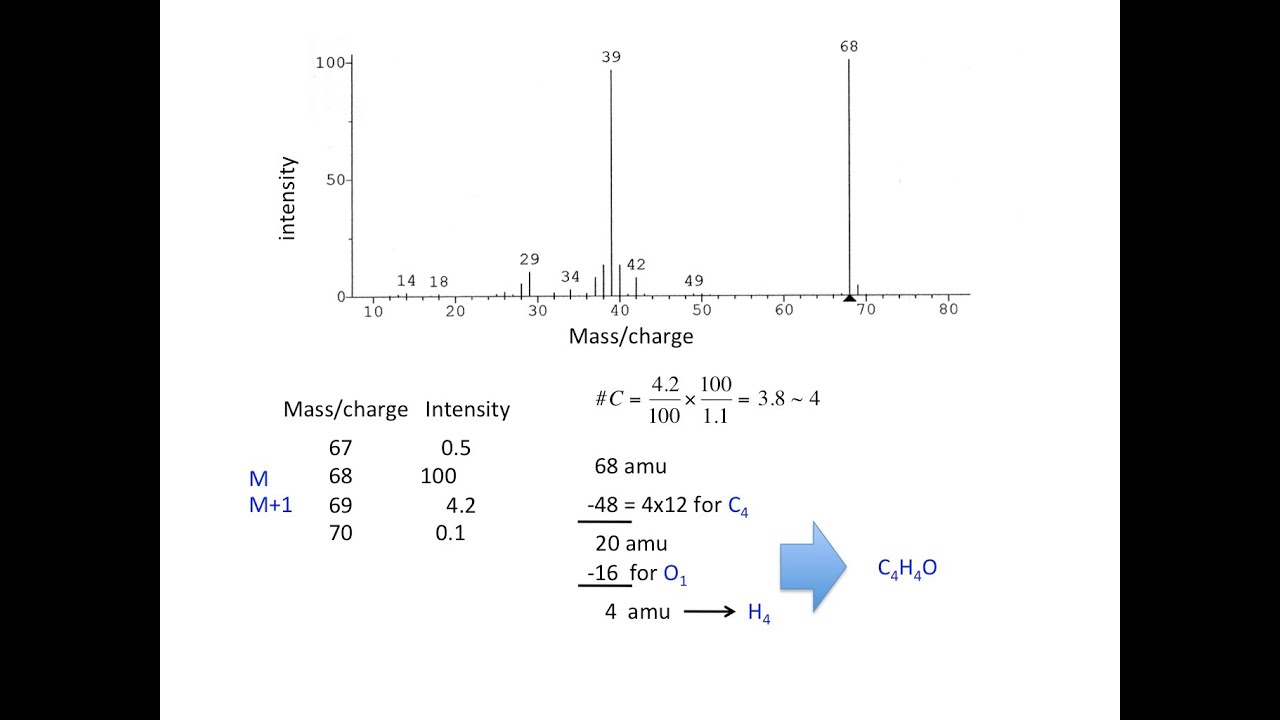
homework - How to calculate m/e value for the base peak in mass spectroscopy? - Chemistry Stack Exchange
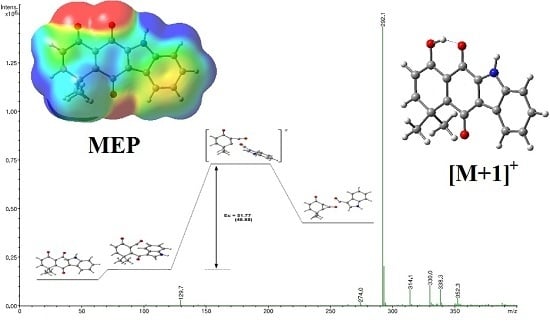
IJMS | Free Full-Text | Protonation Sites, Tandem Mass Spectrometry and Computational Calculations of o-Carbonyl Carbazolequinone Derivatives

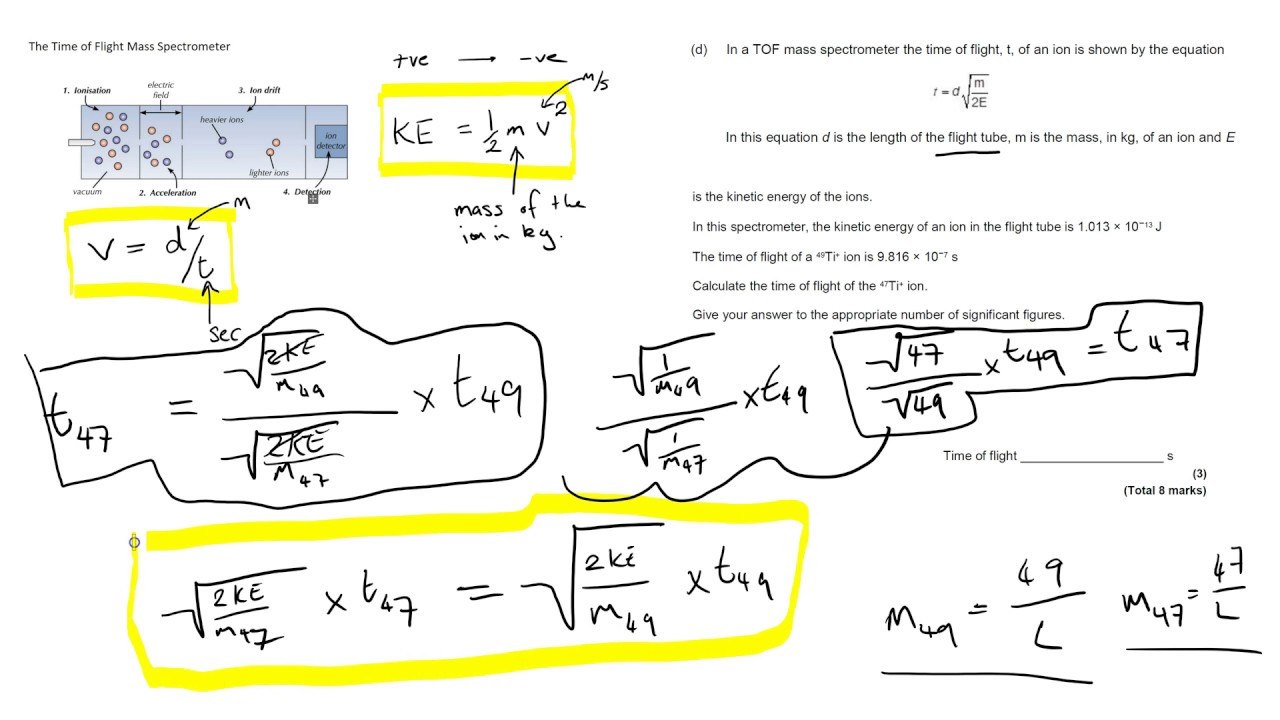
Time of Flight Mass Spectrometry (1.1.3) | AQA A Level Chemistry Revision Notes 2017 | Save My Exams

1.4 THE MASS SPECTROMETER OBJECTIVES: To describe how the mass spectrometer works Make use of mass spectra MAKING SENSE… Try to put what happens in a. - ppt download