
Numerical calculations: Nernst equilibrium potential calculator. The... | Download Scientific Diagram
![SOLVED: 5. Resting membrane potential: a) The Nernst equation allows the calculation of the equilibrium potential of a specific ion Explain how it represents an equilibrium' RT AE = In [Iont oulI[lon"in] SOLVED: 5. Resting membrane potential: a) The Nernst equation allows the calculation of the equilibrium potential of a specific ion Explain how it represents an equilibrium' RT AE = In [Iont oulI[lon"in]](https://cdn.numerade.com/ask_images/eb5407bb9b754701ab11bbb7158bca5b.jpg)
SOLVED: 5. Resting membrane potential: a) The Nernst equation allows the calculation of the equilibrium potential of a specific ion Explain how it represents an equilibrium' RT AE = In [Iont oulI[lon"in]

Using the Nernst equation | Redox reactions and electrochemistry | Chemistry | Khan Academy - YouTube
Write Nernst equation and calculate e.m.f. of the following cell at 298 KPt(s) |Br2l) |Br (0.010 M) ||H (0.030 M) |H2(g) (1 bar) |Pt(s) E(Pt, Br, Br 1.09 V)






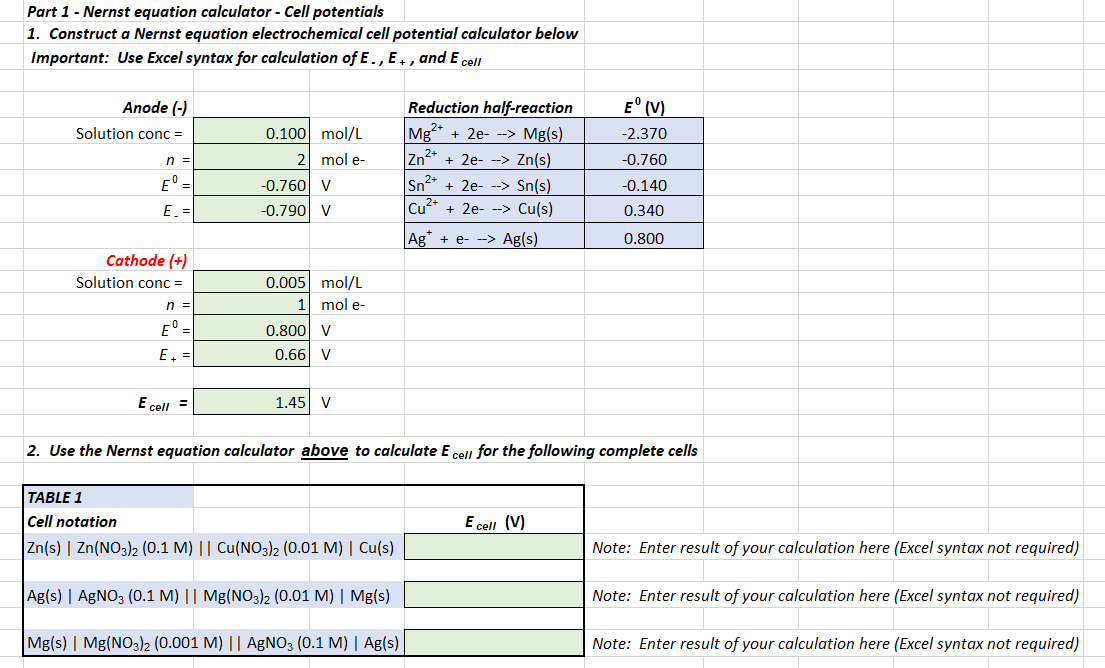







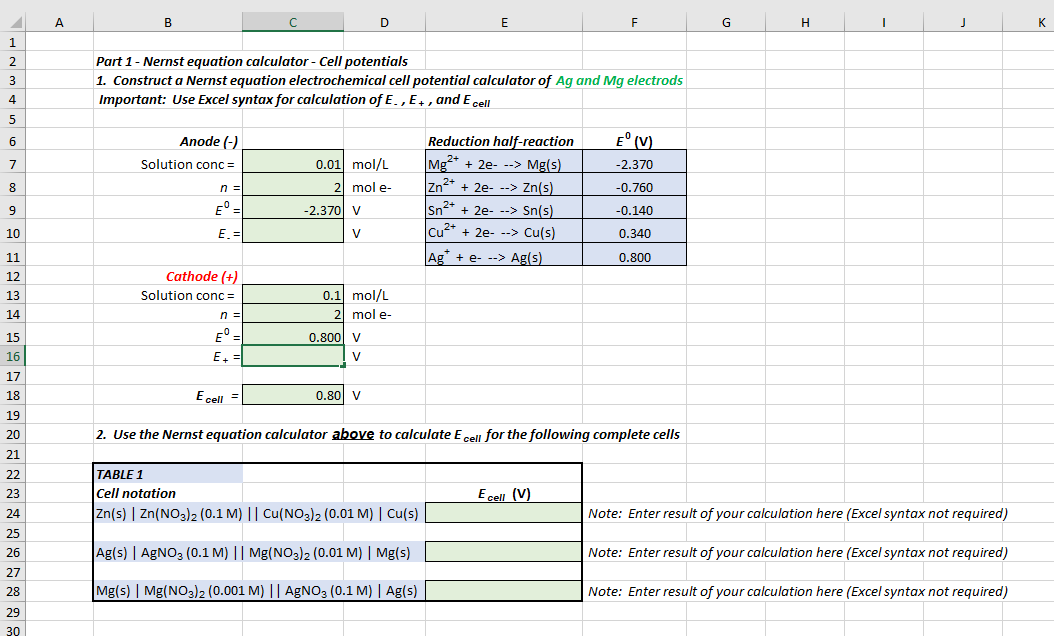

![Solved Part 2 - [Cu“ ] concentration cell Use Nernst | Chegg.com Solved Part 2 - [Cu“ ] concentration cell Use Nernst | Chegg.com](https://media.cheggcdn.com/media/e11/e11d83ac-8412-49cb-b3af-4f1c2271dd7b/phpUIuTCC)
